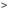Trending Now
- IPL 2024 begins with a bang. First contest between CSK and RCB.
- Election commission allots mike symbol to Naam Thamizhar Katchi
- AIADMK promises to urge for AIIMS in Coimbatore, in its election manifesto.
- Ponmudi becomes higher education minister.
Post
DiabetOmics Launches Lumella® First-of-its-kind Rapid Point-of-care Test for Preeclampsia Assessment
![]() February 7, 2019
February 7, 2019
Preeclampsia, characterized as a hypertensive disorder, is an undesirable complication in pregnancy and can affect both the mother and the baby. It is a complex disease and affects multiple organs and systems in the pregnant woman. If not detected early or not managed in time, severe preeclampsia can result in maternal and infant mortality. Preeclampsia generally occurs after the 20th week of gestation and can sometimes occur even a couple of weeks after child birth. In woman with certain high risk conditions, it is known to occur very early on, in the pregnancy.
 |
L-R: Dr. Pratibha, Dr. Srinivas Nagella, Founder DiabetOmics, Sri. Varaprasad Reddy, Chairman of DiabetOmics,
Dr. Shanthakumari, Dr. Sadasiva Rao
Preeclampsia is a global burden. Incidence rates vary from as low as 2% in the developed countries to as high as 18% in parts of Africa. In Latin America, preeclampsia is the #1 cause of maternal mortality. Ten million women develop preeclampsia each year around the world. Worldwide about 76,000 pregnant women die each year from preeclampsia and related hypertensive disorders. The number of babies who die from these disorders is thought to be on the order of 500,000 per annum. In developing countries, a woman is seven times as likely to develop preeclampsia than a woman in a developed country. 10-25% of these cases will result in maternal death. (Source: Preeclampsia Foundation. www.preeclampsia.org)
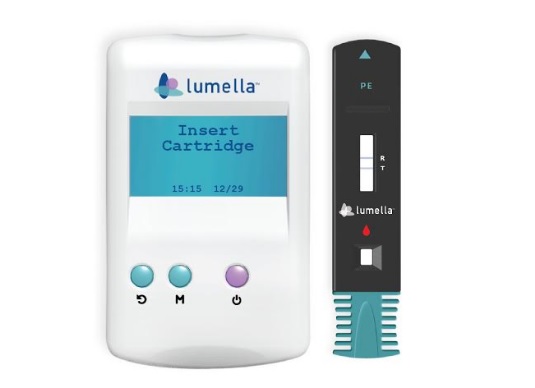 |
Tester and Strip
In India, Preeclampsia is reported to be between 8-10% of the 28 Million pregnancies each year. About 3.5 Million babies are born prematurely in India every year. 45-50% of preterm births are due to unknown causes, 15-20% of these preterm births are due to medically induced or elective pre-term deliveries. (Source: National Health Portal, Govt. of India)
For early detection and timely management of Preeclampsia, antenatal clinics with all facilities capable of dealing with high risk pregnancies, along with trained manpower is an important requirement.
DiabetOmics, a global medical diagnostic company headquartered in Portland, USA, understanding the need for early and reliable assessment of preeclampsia, launched today a new, first-of-its-kind, rapid, point-of-care, test – Lumella. Lumella provides an accurate and reliable estimate of Glycosylated Fibronectin (GlyFN) – a protein biomarker that is elevated in pregnant women with a high risk for developing Preeclampsia. Lumella is a proprietary test developed by DiabetOmics through extensive research. It uses a simple test strip (similar to home pregnancy test kit) and a hand-held quantitative reader. The test can be done with a finger prick blood sample and can be done in the physician’s clinic. Test results are available in 10 minutes and therefore the assessment can be completed within the same visit of the pregnant woman to consult with her Obstetrician. The test being simple and easy to use, enables its adoption in any setting – remote, rural or urban. The Lumella test system, has recently received regulatory approval in India, said Dr. Srinivas Nagalla, Founder & CEO of DiabetOmics Inc.
On the launch occasion, Sri. Varaprasad Reddy, Chairman of DiabetOmics, unveiled their first product, Lumella test for Preeclampsia. The first Lumella test systems were handed over to (1) Dr. Ramesh Kancherla, Chairman & Managing Director of Rainbow Hospitals, Hyderabad, (2) Dr. Swapna Yendru, Director of Swapna Healthcare, Hyderabad, and (3) Dr. Sridhar, Managing Director of an Obgyn Hospital in Kakinada.
Dr. Shanthakumari, Immediate Past Chairperson of Indian College of Obstetricians and Gynaecologists (ICOG) presided over the function and outlined the magnitude of Preeclampsia in India. Dr. Pratibha, Retd. Professor and HOD of Dept of Obgyn, Osmania Medical College, Hyderabad, briefed about the situation in a public health setting.
About DiabetOmics
DiabetOmics, Inc, a global medical diagnostics company pioneering innovative, non-invasive point-of-care tests for diabetes and maternal health was founded in 2009 by Dr. Srinivas Nagalla and Dr. Paturi Vishnupriya Rao.
DiabetOmics Medical, the Indian arm of DiabetOmics, Inc. is a high capacity test strip and reader manufacturing facility located in Medak, Telangana. This state-of-the-art facility with a capacity to manufacture 5 million tests a year is ISO 13485 certified with necessary regulatory approvals and is fully geared up to meet global sales requirements of Lumella. DiabetOmics is also supported by Technology Development Board, Department of Science & Technology, Govt. of India for development and commercialization of its range of novel point-of-care tests including Lumella.
Lumella is the first product from DiabetOmics for the Obstetrics and Gynecology specialty, and has a pipeline of other novel, in-vitro diagnostic products targeted at affordable, reliable and non-invasive diagnosis and management of Diabetes.
DiabetOmics has been awarded six key patents and prosecuting both domestic and international patents designed to protect its ownership of the diabetes diagnostic market based on its non-invasive and point-of-care technology.